
“Quality improvement methodologies are frequently used to improve health care delivery, but can also be leveraged to improve research processes. If your study team is facing a research barrier, we can help you to select and use the appropriate tools and strategies to overcome it.”
Denise Daudelin, RN, MPHDirector, Research Process Improvement
Overview
The Center for Research Process Improvement builds the capacity of research teams to conduct efficient, cutting-edge research across the translational spectrum. This program helps researchers use methods already proven in industry and health care to increase efficiency and quality of research by addressing challenges and barriers in the research process.
Process improvement methods include a systematic approach to resolving problems and reducing barriers. The following list contains some examples of quality improvement tools and why a researcher might use them:
- Process Mapping: Helps teams understand the steps involved in a process.
- Failure Modes and Effects Analysis (FMEA): Identifies possible failures in a process and allows team to rank steps based on severity, likelihood of occurrence, and likelihood of detection.
- Cause-and-effect Diagram: Clarifies the underlying causes of a problem.
- Key Driver Diagram: Visually displays conditions that need to be met in order to achieve a project aim. Includes brainstormed change ideas.
- Plan-Do-Study-Act (PDSA) Cycles: Iterative tests of change that allow teams to start small and gradually increase the size of their test as their confidence in the change builds. These rapid cycles are ideal to receive quick feedback before a team is ready to implement the change system-wide.
- Runcharts: Captures data over time in the form of a line graph. Using statistical rules, it can help teams decide what is common cause variation and what is a change that is likely attributable to the idea tested. Also allows teams to track their progress over time.
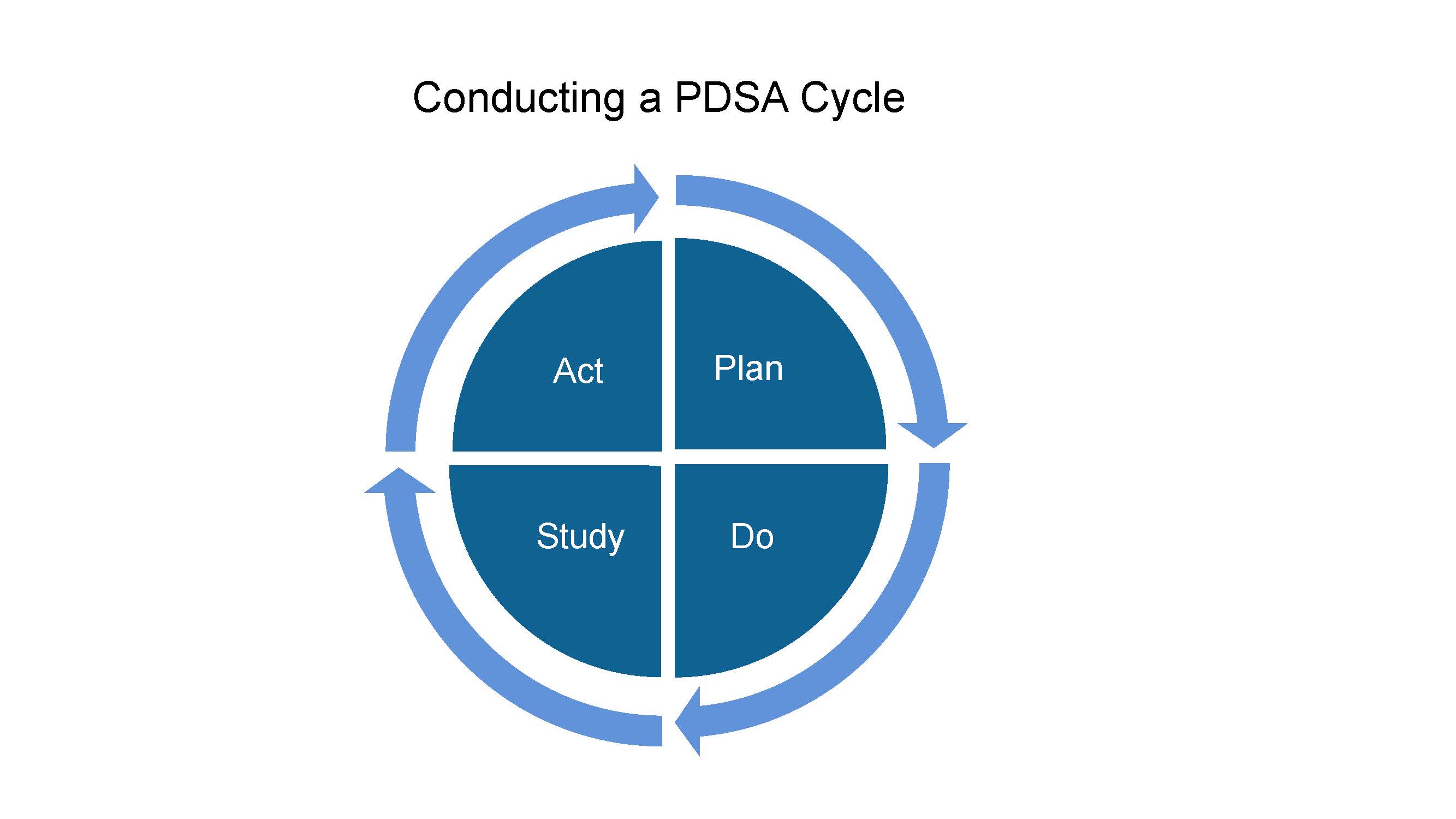
Conducting a PDSA Cycle
Description of the Plan-Do-Study-Act (PDSA) Cycle diagram
Here are the steps to conduct a Plan-Do-Study-Act (PDSA) Cycle:
- Plan the change your team will test. Who will do what and by when? What do you expect will happen?
- Do the test.
- Study the results. Did you achieve your expected result? Why/why not?
- Act. Decide if you’ll adopt, adapt, or abandon this change for the next cycle.
Who is eligible?
The Research Process Improvement Program helps individuals and teams to improve research processes and offers consultations on using improvement methods in research studies. Faculty and trainees at Tufts CTSI partner and collaborator institutions are eligible for these consultation services.
Services
Training, consultation, and coaching are available to career development awardees, investigators and research teams. Services are tailored to the needs of each research team. In addition, our interactive education website, I LEARN, includes several RPI seminars to get you started using improvement methods right away.
Download and share our flyer (PDF).
Resources
The Research Process Improvement Toolkit (PDF) is now available! The Toolkit highlights quality improvement tools that can be used in research. Worksheets and templates are included to assist research teams with their improvement projects.
The Study Start-up Toolkit walks research staff through the steps of getting an industry-sponsored study ready to enroll participants. In this guide, you will find resources, checklists, an IRB decision tool, and a process map to demystify the process. The guide is available on the intranet and through this link.
Wondering whether you should conduct a survey? Should I Conduct a Survey? (PDF) one-page resource.
Review our Case Studies on How to Improve Clinical Recruitment Rates and our clinical trial metric dashboard, Collecting and Using Clinical Trial Metrics to Improve Trial Performance.
To request RPI assistance, please complete our service request form.
Contact
To contact us, please submit a service request.
Denise Daudelin, RN, MPH
Director, Research Process Improvement
Alyssa Cabrera, MPH
Senior Project Manager



